What research can be done to investigate EFHC1 and Epilepsy?
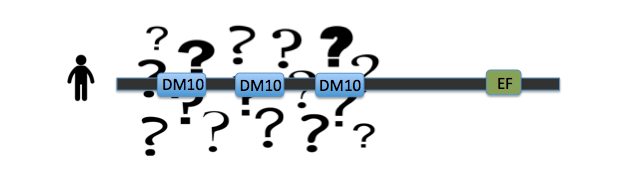
The largest problem that remains in the investigation of EFHC1's role in juvenile myoclonic epilepsy is the domain functions of the protein. While the EF hand has been documented to be involved in calcium binding, the triple repeat DM10 domains remain of unknown function [1]. And it is within these DM10 domains that the disease-causing mutations are occuring [2]. Without a basic understanding of the functions of the protein itself, our understanding of the causality of epilepsy remains incomplete, and treatments are aimed at the symptoms, not at the cause of the disease. By investigating the roles of these DM10 domains in protein function, new options for treatment could arise to stop the problem at the source.
|
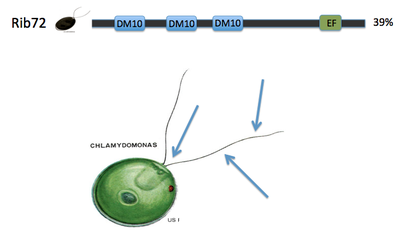
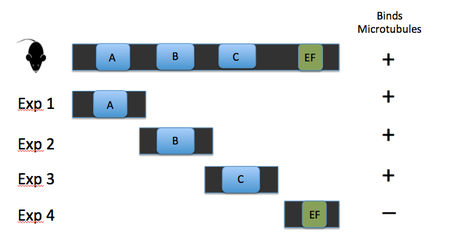
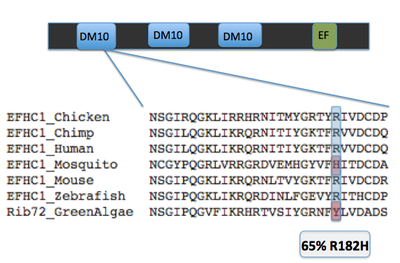
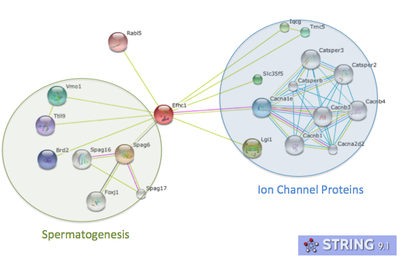
Key evidence for the role of DM10 domains in EFHC1 function comes from studies of Rib72, the homolog of EFHC1 of chlamydomonas, or green algae single cellular organisms (see image 1). Because green algae have no nervous system, and no neurons, the protein does not have a role in neuronal firing. However research has proven its essential role in flagellum structure [3]. Rib72 binds to microtubule structures, forming protofilaments within the flagellum of the algae [3]. To test this hypothesis, I have several suggestions for future studies. The first is a structure-function analysis of the EFHC1 protein, as depicted in image 3. This test involves chopping the protein in order to isolate each individual DM10 domain, here labeled A B and C. These individual domains will then each be tested for their ability to bind microtubule units by tagging microtubules with a fluorescent tag. This test will give detail to which specific DM10 domains are able to bind microtubules, or if the DM10 domains are incapable of binding in isolation. My next experiment would be to investigate in other organisms the amino acid changes that cause the epilepsy state in humans. I would predict that the amino acids so vital to EFHC1 function in humans would also be vital to protein function in homologous proteins. The first of these experiments was conducted using ClustalOmega sequence alignment and observing the epilepsy genotype of Argenine 182 transforming to Histidine. This argenine is largely conserved throughout homologous proteins, however both mosquito and green algae contain a different amino acid at that location. These are the farthest phylogenetic relatives of humans from the list given so it is not surprising that these two organisms have a different residue. However, it is surprising that mosquitos contain a histidine at the 182 location, the very amino acid that causes the disease state for humans [4]. My third experiment would be to investigate the protein interaction partners of EFHC1, as depicted in image 5 sorted into proteins of general similar function. I would search for proteins within this web that are involved in microtubule synthesis or regulation as that would provide support for EFHC1's role in microtubule structure. My preliminary analysis of these proteins yielded two domains of important function to cellular microtubules. Tektin1 protein contains a tektin domain which forms protofilaments of microtubules in flagellum, much like our Rib72 protein [5]. Tt119 also contains a microtubule associated domain - tubulin-tyrosine kinase domain - which conducts post-translational modification of microtubules during synthesis [5]. |
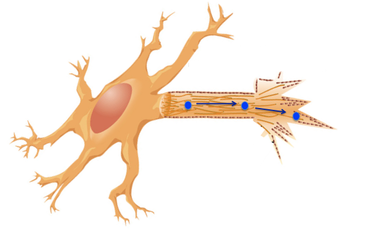
The green algae EFHC1 homolog having an involvement in microtubule structure is consistent with our EFHC1 causation of epilepsy. Neurons contain vast microtubule structures serving as an information highway along which messages can be transported (see image 2). When EFHC1 is mutated and non-funtional, these microtubule structures could be irregular leading to inappropriate neuronal firing. With this idea in mind, my hypothesis for future research is that EFHC1 binds microtubules using the DM10 domains within the protein. These experiments would provide support for my hypothesis of DM10 domain's involvement in microtubule structures. A further question I have in my research concerns sterility among juvenile myoclonic epilepsy patients. My search through JME research found no studies that yielded results of increased sterility in JME patients with EFHC1 mutations. This is surprising given the role in microtubule efficacy EFHC1 appears to play. Wtihout effective microtubules, the flagella of sperm and cilia of the fallopian tubes would have decreased effectivity, increasing reproductive problems for JME patients. I would conduct an extensive study of living JME patients to analyze the microtubule structures within these reproductive organs to look for signs of increased sterility due to EFHC1's effects. |
| victoriavanvreedeefhc1pres.pptx | |
| File Size: | 2676 kb |
| File Type: | pptx |
References: [1] De Nijs et. al., Juvenile myoclonic epilepsy as a possible neurodevelopmental disease: Role of EFHC1 or Myoclonin1. Epilepsy and Behavior. Volume 28. July 2013. http://www.sciencedirect.com/science/article/pii/S1525505012004969
[2] Delgado-Escueta et. al. The quest for Juvenile Myoclonic Epilepsy genes. Epilepsy and Behavior Journal. Volume 28. July 2013. http://www.sciencedirect.com/science/article/pii/S1525505012004957
[3] King, Stephen. Axonemal Protofilament Ribbons, DM10 Domains, and the Link to Juvenile Myoclonic Epilepsy. Cell Motility and the Cytoskeleton. Volume 63. 2006. http://onlinelibrary.wiley.com/store/10.1002/cm.20129/asset/20129_ftp.pdf?v=1&t=hturpcy2&s=ac620ef3194976e78700d12e0d250a790d2c6f20
[4] Jara Prado et al. Novel Myoclonic/EFHC1 mutations in Mexican patients with juvenile myoclonic epilepsy. Seizure Journal 2012.
[5] Smart Database http://smart.embl.de/
Images:
http://en.wikipedia.org/wiki/File:Kinesin_cartoon.png
http://string-db.org/newstring_cgi/show_network_section.pl
http://www.newconceptinfosys.com/ibm/imagegalerysecond.php?imageID=2
http://zaraweir.wordpress.com/http://7bigspoons.com/fertility-2/thankfully-sperm-dont-directions/
http://www.chp.edu/CHP/juvenile+myoclonic+epilepsy
http://www.ncbi.nlm.nih.gov/homologene